The following is from MedPage Today.
The FDA added a boxed warning to the label of the osteoporosis drug denosumab (Prolia) over the risk of severe hypocalcemia in patients with advanced chronic kidney disease (CKD), the agency announced Friday.
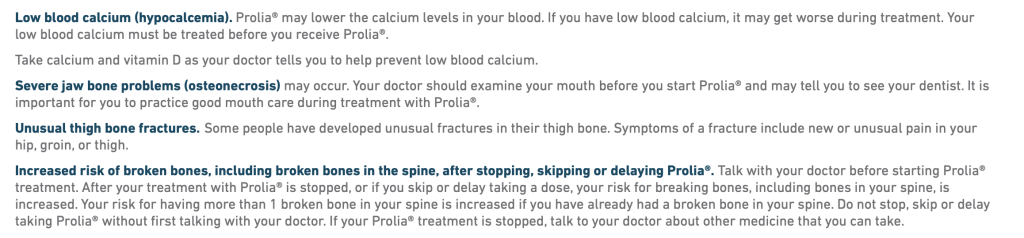
After a review of the evidence, including new data published in JAMA, the FDA determined that denosumab can increase the risk for severe hypocalcemia compared with bisphosphonates in patients with advanced CKD, especially those on dialysis.
Patients on dialysis or with mineral and bone disorder (MBD) have the highest risk of very low blood calcium levels.
The new data, which involved women on dialysis, showed that 41% developed severe hypocalcemia during the first 12 weeks of denosumab treatment, compared with 2.0% of those taking oral bisphosphonates.
The FDA’s “most prominent” warning, the boxed warning lists the potential for serious harm from severe hypocalcemia. This could include hospitalization, life-threatening events, and death in patients with advanced CKD taking denosumab.
Severe hypocalcemia can also be asymptomatic or can present with symptoms like confusion, seizures, irregular heart rhythm, fainting, face twitching, uncontrolled muscle spasms, or weakness, tingling, or numbness in parts of the body.
As part of its new drug safety communication, the FDA advised clinicians to carefully select appropriate patients for this treatment and increase monitoring of blood calcium levels, particularly for the first 2 to 10 weeks after each injection. Calcium and vitamin D supplements may also be required. (Continued…)
Link to article here.

Visit The Sharyl Attkisson Store today
Unique gifts for independent thinkers
Proceeds benefit independent journalism



